
insights
Managing Clinical Trials So They Don’t Break the Bank: Careful Planning Increases Efficiency
I recently partnered with Laurie Halloran, President & CEO of Halloran Consulting Group, to share our perspective on “Managing Clinical Trials Creatively and Efficiently So That They Don’t Break the Bank” at a recent Biopharma Hub virtual event. The focus of the presentation was to provide attendees with some concrete examples of meaningful steps that can be taken to reduce the cost and time it takes to conduct clinical trials and ultimately improve the odds of getting a new drug to market. Our presentation covered several angles:
- Big picture on costs and risk
- Thoughtful approach to integrated development planning
- Examination of individual activities within clinical study stages:
- Study planning
- Study startup
- Study conduct
- Study closeout
Big Picture on Costs and Risk: The Problem
To provide some hard numbers, on average, it takes about 10-15 years to bring a compound from the lab to market at an overall cost of $500 million to $2.6 billion. Approximately one out of every 1,000 of these compounds make it from lab to clinical stage, and less than 0.1% make it all the way from the lab to market. The clinical stage of development is the riskiest and most costly, taking between 6.7-7.5 years at a cost of up to $1.5 billion. Less than 12% of drugs that enter the clinical stage ultimately become marketed drugs. With numbers of this magnitude, even small changes to clinical programs can have major impacts to a manufacturer’s bottom line.
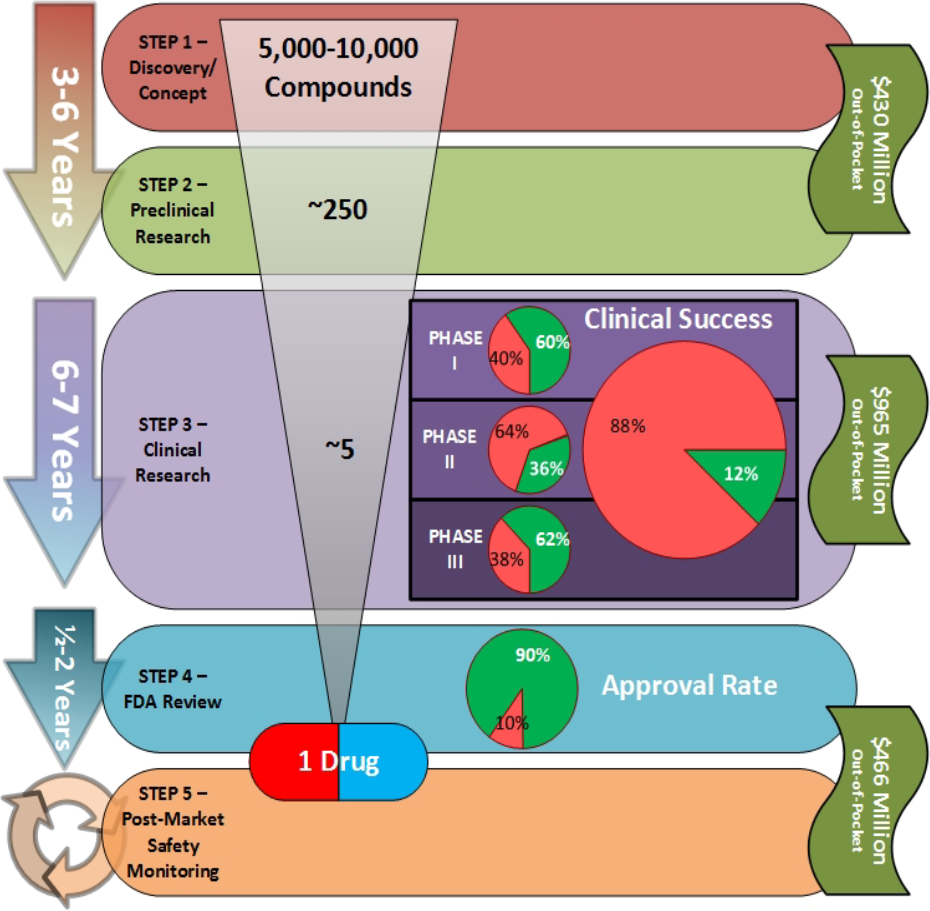
Figure 1 – Drug Development Pipeline. Slagle, B. M. (2018). An Evaluation of Global Indicators as Surrogates for Clinical Research Capacity in the US, Poland, and Vietnam [Master’s Thesis, Harvard University]. http://nrs.harvard.edu/urn-3:HUL.InstRepos:37945112
Recommendations and Solutions
A common theme throughout the discussion, and perhaps not surprisingly, was that careful planning up front pays off down the line. We started with the 30,000-foot view then dove down to ground level to walk through both high-level and focused strategies to increase clinical trial efficiency.
A Mindful Approach to Integrated Development Planning
Perhaps the most impactful step a company developing a new drug can take to increase the efficiency of their clinical program is taking the time in the early stages of development to develop an integrated development plan (IDP). This requires stepping back as a team to map out individual product development activities (e.g., non-clinical, CMC, regulatory, clinical, etc.) based on key assumptions, then weaving those plans together into one large IDP. It’s important to perform this step prior to entering the clinical stage to ensure the development pipeline is strategically positioned to minimize risk and increase the probability of success.
There’s a lot of work that goes into planning, but if well executed, this mindful approach can save organizations from costly mistakes that could have been prevented with more time upfront during the planning and diligence phase. We’ve also identified many advantages of diving deep into the plan, and the detailed preparation often leads to:
- A detailed outline of major development milestones with successful criteria defined
- An understanding of cost and timeline estimates that provide the basis for investment decisions
- A data-driven portfolio prioritization and management
- Reduction of surprises by thinking through various scenarios in detail
- Assessment of most efficient and cost-effective pathway to market
The level of detail captured in the IDP can vary depending on the maturity of the asset, and it should be regularly revised to account for the evolution of various activities.
IDP Considerations for Various Stages of Maturity
For earlier stage assets, the IDP should be fairly broad, focusing on the minimum steps required to make a regulatory submission based on historical benchmarks for key activity timelines and costs. The high-level IDP should be developed in parallel with other strategic activities, such as indication assessment prioritization, interviewing Key Opinion Leaders (KOLs), and developing a competitive intelligence and landscape assessment of the product to evaluate how successful it will be amongst competitors.
Developing an IDP requires cross-functional alignment between stakeholders from clinical, CMC, regulatory, toxicology, and others. Teams will need to lean on each other’s expertise to discuss scenarios to identify risks and mitigation, access a high-level market assessment and identify commercial viability, and evaluate the probability of technical regulatory success (PTRS). The collaboration of these team members is crucial to look at product development activities from all angles, and to understand how changes to one step can impact downstream dependencies.
As the asset evolves the IDP then begins to focus on how each activity will be done with vendors, timelines, and costs. The IDP can be coupled with stage gates and inflection points to determine when to change course or even terminate the development of an asset based on Net Present Value (NPV) and PTRS calculations.
In summary, taking the time to develop detailed IDPs early in the development process results in significant efficiency gains throughout. Planning through and de-risking various scenarios on the front end prevents overrunning costs and timelines due to oversights and inefficiencies on the back end.
Individual Study Level – Planning Stage
There are also a number of steps that can be taken at the individual study level to increase efficiency. To keep in step with the previous section, these have the biggest impact when thoroughly considered during study planning stages rather than when a study is over budget and running over time. Some examples of planning stage steps that can be taken are as follows:
- Lean Protocol Design – Think thin! Take a critical eye to the study visits and assessments and trim wherever possible so only the critical endpoints are included. In one example, by convincing a client to remove an unnecessary diffusing capacity of the lungs for carbon monoxide (DLCO) assessment for a COVID-19 study they saved over $1.6 million.
- Incorporate DCT Elements Whenever Possible – On-site visits require lots of resources and add to participant burden. By allowing for some visits to be virtual, millions can be saved throughout the course of a study and reduce the burden on participants.
- Thorough Protocol Feasibility – The realization that a protocol has major procedural gaps once the study launches, and sites identify issues is all too familiar. Take some extra time to work with cross-functional stakeholders, PIs/KOLs, site staff, and patient advocacy groups to work the kinks out of a protocol before finalizing is a big undertaking, but it’s worth it. For a phase 2 study, the cost of an unnecessary amendment is over $150k; for a phase 3 study it’s well over $500k.
- Thoughtful Site Selection – Don’t rely on KOLs to deliver participants based on their credentials and overly ambitious projections. Consider a mix of large and small sites of varying levels of experience. A smaller site may require a bit more hand-holding but are often eager to make a name for themselves and exceed expectations.
- Fit-For-Purpose Systems/Technology – Think twice about using advanced eCOA/ePRO and high-end technologies for an early-stage study. Are they really necessary or just shiny? On the flip side, consider how the data will be used with the next study. Is it worth picking a mid-tier EDC system rather than the cheapest option?
- Fit-For-Purpose CROs/Vendors – There are pros and cons to using big CROs as well as using smaller CROs/vendors. Take the time to consider the needs of a study and if it makes sense to leverage bundled services at a higher overall cost versus multiple individual vendors with lower individual costs. Think about the resources needed to manage several individual vendors.
Individual Study Level – Start-Up Stage
Once the study is designed and ready to launch, there are a few more opportunities to cut costs. A couple of examples include:
- Virtual Investigator Meeting – Everyone loves an in-person investigator meeting, and you can’t beat a face-to-face interaction, but there may be other ways to build rapport. Prioritize sponsor representation at SIVs to make stronger connections with each site. It could also save you over $100k off the study budget.
- Finalize Documents Prior to Training – Training study teams, vendors, and sites to the protocol and study documents is a critically important step, and there is a tendency to hold onto the first way you learn to do something (primacy). So, if training to study plans, manuals, etc. is rushed and the documents are changed afterward, there WILL be compliance issues when you ask people to train to revised versions. Take some extra time to make sure documents are quality controlled and finalized before releasing them.
Individual Study Level – Conduct Stage
Congratulations! You made it through start-up, got your first sites up and running, and are now enrolling participants across the globe. It’s smooth sailing from here, right? Here are a couple of pointers, just in case:
- CRO/Vendor Oversight – Don’t sit back and expect everything to stay on course on its own. CRO/vendor oversight/management is one of the most important tasks for a sponsor. Work out KPIs, metrics, and custom reports early on and review them in detail regularly. Additionally, CRO/vendor turnover is inevitable. Work with a CRO/vendor to ensure there is robust documentation in study plans for succession planning and that detailed transition plan templates are available at the start of the study.
- Monitoring – “Implementing RBM isn’t worth it because it doesn’t save that much money,” is a comment heard often. While RBM doesn’t always result in a smaller monitoring budget, it does identify potential site issues faster and more accurately than traditional 100% SDV monitoring. This can reduce the cost of dealing with compliance and data issues in the long term. Work smarter, not harder.
Individual Study Level – Closeout Stage
Even after LPO – the date and time when the last patient has reached a planned or achieved milestone representing the completion of the trial – and DBL – the finalization of data sets obtained in a clinical trial – there are still chances to save some money.
- Take Things In-House – As things wind down and there are only a few participants in long-term follow-up, evaluate the in-house team’s capacity for taking over activities from CROs and vendors. As this question ‘can your in-house CTM/CTA take over eTMF management or IP reconciliation?’
Conclusion
Some of the biggest cost savings result from careful up-front planning. Whether at the individual study level or across multiple functions and programs, taking extra time to plan through the details on the front end beats trying to make up for oversights on the back end.
**
Contact us to learn more about Halloran Consulting Group’s Clinical Services and how we assist companies like yours from early drug development to commercialization.
About the Author
Brendan Slagle, Senior Consultant, Clinical Development, is passionate about helping to bring life-changing therapies and products to market, while ensuring their safety and efficacy through robust clinical programs. His key areas of expertise include the European Medical Device Regulation, Post-Market Clinical Follow-up strategy and plan design, Clinical Evaluation Reports, and strategic publication planning. Reach out to Brendan by email or through LinkedIn.




